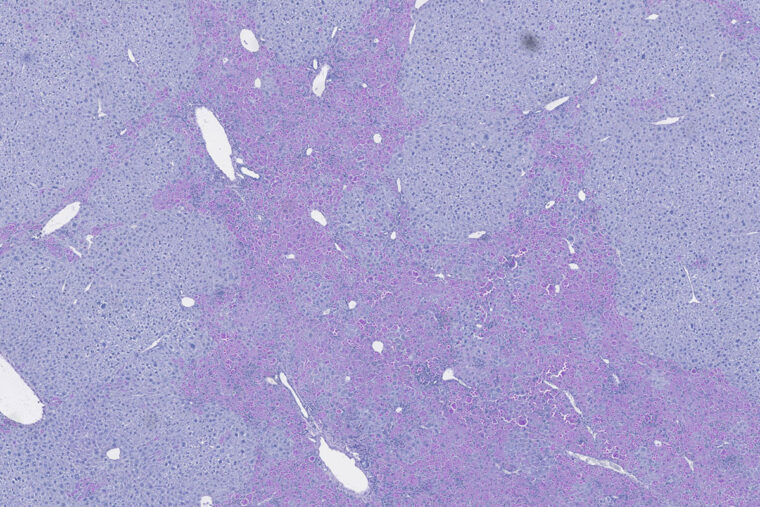
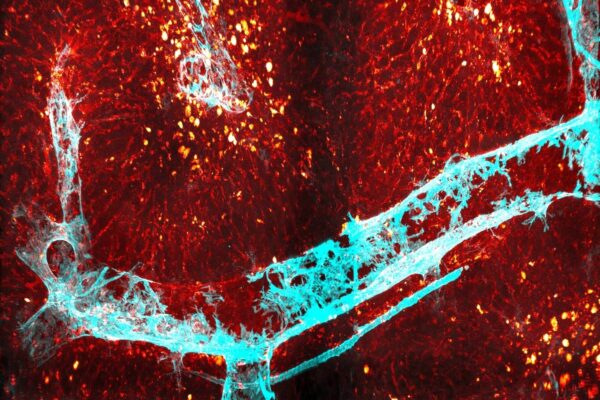
Alpha1-antitrypsin deficiency, an inherited disorder affecting 100,000 people in the U.S., causes a progressive and incurable lung disease. A subset of patients with the condition — about 10% to 15% — also develop liver disease because of the accumulation of the aggregated protein variant resulting from the genetic error that causes the disease. Now, researchers at Washington University School of Medicine in St. Louis have identified a previously unknown biological process that helps explain why only a subset of the affected population develop liver disease.
The newly discovered process appears to protect liver cells from the toxic effects of the buildup of misfolded, aggregation-prone proteins inside those cells. The findings could help clarify the wide variation in disease severity sometimes seen among patients with alpha1-antitrypsin deficiency and inform new approaches to predicting which patients are at highest risk of eventually needing a liver transplant.
The study appears online in the journal Nature Communications.
Cells have tightly controlled processes to make all the proteins necessary for healthy functioning. There are processes for manufacturing proteins, for folding them into their proper shapes, for transporting them to the correct parts of the cell, and for degrading and disposing of them when they’re no longer needed. Because disruptions in any part of this process — called proteostasis — can lead to disease, cells also have safety measures to handle mistakes that crop up along the way.
“What is truly remarkable about proteostasis is that it’s set up to have multiple fail-safes for handling a bad protein,” said senior author David H. Perlmutter, MD, executive vice chancellor for medical affairs, the Spencer T. and Ann W. Olin Distinguished Professor and the George and Carol Bauer Dean of WashU Medicine. “That’s good for cellular economy because it means the cell doesn’t have to spend all its energy on making every protein perfectly. Our study identifies a completely new way that cells manage potentially harmful proteins.”
Perlmutter and his colleagues dubbed the newly identified fail-safe the polymerized protein response. The new study suggests that this response allows cells to tolerate misfolded proteins that also polymerize and aggregate, maintaining normal cell function despite their presence. It complements a separate, well-characterized quality-control process, the unfolded protein response, that governs how cells handle unfolded proteins.
Both unfolded proteins and aggregated proteins can accumulate in a cell’s endoplasmic reticulum — the protein manufacturing, packaging and shipping center of cells. And the new study now shows that cells have different processes for handling each.
Studying human cell lines and mouse models of alpha1-antitrypsin deficiency, Perlmutter and his colleagues showed that aggregated proteins in the endoplasmic reticulum of liver cells trigger the polymerized protein response through a molecule called Derlin-2, which then activates an important molecule called an NF-kappa-B p50 homodimer. These interactions set in motion a genetic program that protects the cell. Perlmutter and his team are continuing this research to work out the details of that protection.
Perlmutter said the identification of this new process could help researchers design ways to identify which patients with alpha1-antitrypsin deficiency are at highest risk of developing severe liver disease and will likely require a liver transplant. Early identification — before the liver damage becomes apparent — could help guide treatment decisions and reveal potential routes to new therapies and prevention strategies.
“We think the polymerized protein response is a cellular adaptation that protects most patients from liver damage due to these aggregated proteins building up in their liver cells,” Perlmutter said. “It allows the cells to be healthy despite the presence of the proteins. As long as this signal is present, the cells are able to handle the protein load.”
Importantly, the study found that the polymerized protein response could apply to aggregated proteins in diseases beyond alpha1-antitrypsin deficiency. Their experiments showed that the response can be triggered in a rare age-dependent dementia, inherited diabetes insipidus and amyotrophic lateral sclerosis (ALS) because each of these is caused by a genetic variant that aberrantly polymerizes in the endoplasmic reticulum of the cells in which they are made.
“We are continuing to investigate the molecular details of the polymerized protein response and how it plays a role in a host of diseases caused by aggregation-prone proteins, in addition to alpha-1 antitrypsin deficiency,” Perlmutter said.
Munanairi A, Rudnick DA, Huang J, Perlmutter DH. The polymerized protein response (PPR) is activated by genetic variants that polymerize in the ER and is mediated by NFkappaBp50. Nature Communications. April 27, 2026. DOI: 10.1038/s41467-026-72369-w.
This work was supported by the National Institutes of Health (NIH) through U.S. Public Health Service grants P01-DK096990 and R01-DK131215. Additional support was provided by the Genome Engineering and Stem Cell Center at Washington University in St. Louis; the Genome Technology Access Center at WashU Medicine; researchers at Washington University School of Medicine in St. Louis; and researchers at the University of Basel, Switzerland. This content is solely the responsibility of the authors and does not necessarily represent the official view of the NIH.
About WashU Medicine
WashU Medicine is a global leader in academic medicine, including biomedical research, patient care and educational programs with more than 3,000 faculty. Its National Institutes of Health (NIH) research funding portfolio is the second largest among U.S. medical schools and has grown 83% since 2016. Together with institutional investment, WashU Medicine commits well over $1 billion annually to basic and clinical research innovation and training. Its faculty practice is consistently among the top five in the country, with more than 2,000 faculty physicians practicing at 130 locations. WashU Medicine physicians exclusively staff Barnes-Jewish and St. Louis Children’s hospitals — the academic hospitals of BJC HealthCare — and Siteman Cancer Center, a partnership between BJC HealthCare and WashU Medicine and the only National Cancer Institute-designated comprehensive cancer center in Missouri. WashU Medicine physicians also treat patients at BJC’s community hospitals in our region. With a storied history in MD/PhD training, WashU Medicine recently dedicated $100 million to scholarships and curriculum renewal for its medical students, and is home to top-notch training programs in every medical subspecialty as well as physical therapy, occupational therapy, and audiology and communications sciences.
Originally published on the WashU Medicine website