Plastics are ubiquitous in our society, found in packaging and bottles as well as making up more than 18% of solid waste in landfills. Many of these plastics also make their way into the oceans, where they take up to hundreds of years to break down into pieces that can harm wildlife and the aquatic ecosystem.

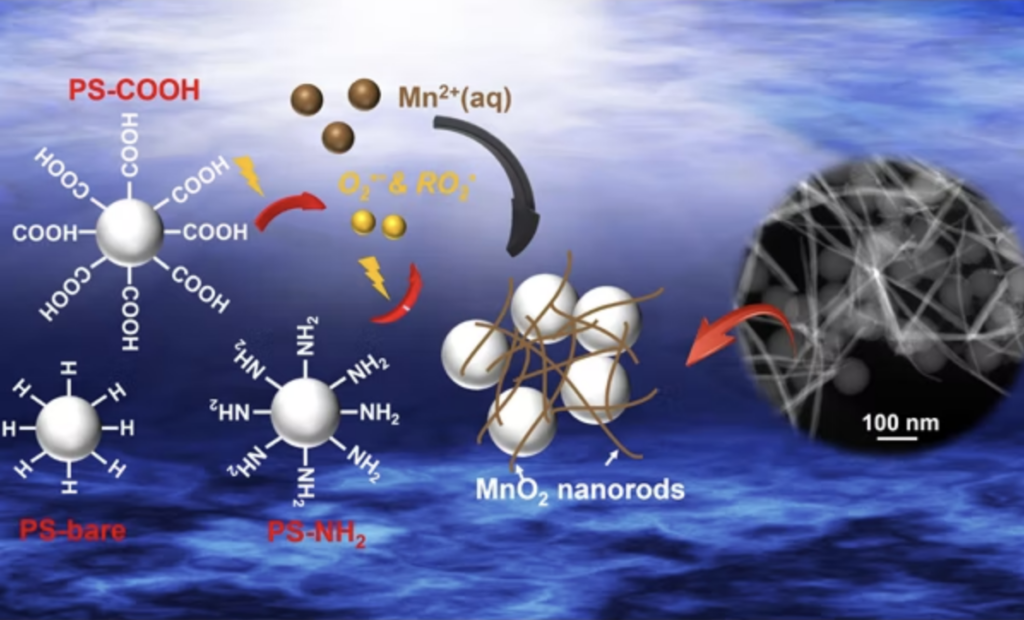
A team of researchers, led by Young-Shin Jun, a professor of energy, environmental and chemical engineering at the McKelvey School of Engineering at Washington University in St. Louis, analyzed how light breaks down polystyrene, a nonbiodegradable plastic from which packing peanuts, DVD cases and disposable utensils are made. In addition, they found that nanoplastic particles can play active roles in environmental systems.
In particular, when exposed to light, the nanoplastics derived from polystyrene unexpectedly facilitated the oxidation of aqueous manganese ions and formation of manganese oxide solids that can affect the fate and transport of organic contaminants in natural and engineering water systems. The research was published Dec. 27 in ACS Nano.
“As more plastic debris accumulates in the natural environment, there are increasing concerns about its adverse effects,” said Jun, who leads the Environmental Nanochemistry Laboratory. “However, in most cases, we have been concerned about the roles of the physical presence of nanoplastics rather than their active roles as reactants. We found that such small plastic particles can more easily interact with neighboring substances, such as heavy metals and organic contaminants, and can be more reactive than we previously thought.”
Jun’s team plans to study the breakdown of diverse common plastic sources that can release nanoplastics and reactive oxidizing species and to investigate their active roles in the oxidation of transition and heavy metal ions in the future.
Read more on the McKelvey School of Engineering website.
Gao Z, Chou P-I, Liu J, Zhu Y, Jun Y-S. Oxidative Roles of Polystyrene-Based Nanoplastics in Inducing Manganese Oxide Formation under Light Illumination. ACS Nano, Dec. 27, 2022. https://doi.org/10.1021/acsnano.2c05803.
Funding for this research was partially provided by the National Science Foundation (CHE-1905077) and the McDonnell International Scholars Academy at Washington University in St. Louis.