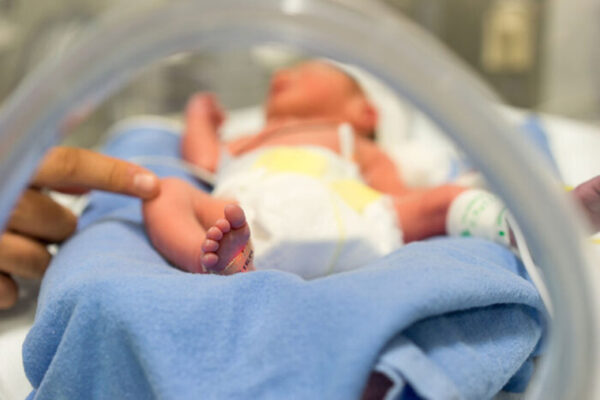
In the dimly lit neonatal intensive care unit (NICU) at St. Louis Children’s Hospital, where Washington University School of Medicine in St. Louis physicians treat the tiniest patients, babies sleep amid the soft hum of machines and gentle whispers of nurses. Doctors and nurses move quickly but with care to avoid disturbing their delicate charges.
Part of protecting these vulnerable infants also means minimizing unnecessary medical interventions. For years, nearly all preemies in U.S. hospitals received prophylactic antibiotics at birth to prevent early-onset sepsis, a bloodstream infection that can occur within the first 72 hours of life. This life-threatening condition can begin in utero or during delivery, and it can quickly become severe. More available antibiotics and earlier use helped reduce neonatal sepsis deaths from 30–50% in the 1960s to 5–20% today. They were long seen as relatively harmless, with no downside, and thus used widely and for extended periods of time.
Then an era of antibiotic stewardship around the turn of the century ushered in global efforts to avoid unnecessary antibiotic use to combat the rise of dangerous pathogens that can evolve to outsmart antibiotics’ killing powers. Researchers at WashU Medicine have added to this effort through studies revealing an important downside to universal antibiotic use in preemies: It can cause long-lasting damage to the developing communities of microbes in the babies’ intestinal tracts, known as the gut microbiome, and might paradoxically increase the risk of subsequent bloodstream infections.
As a result of these findings, antibiotic use in the NICU at St. Louis Children’s is now reserved for those babies who have suspected infections, a practice that has reduced the use of the drugs from nearly 100% of preemies to 70%, without increasing mortality rates. WashU Medicine researchers are working to further reduce antibiotic use in the NICU by developing tools that can help physicians either identify or calculate the risk of an infection faster in preemies.
“The decision to use — or not use — antibiotics in premature infants is still an imperfect science,” said Barbara Warner, MD, a professor of pediatrics and director of the Division of Newborn Medicine at WashU Medicine who has led several studies of the gut microbiome in preemies. “But by carefully assessing each case and tailoring treatment, we have made significant strides to prevent unnecessary antibiotic exposure.”
Care backed by research
Each year, more than 1,700 babies receive specialized care in the St. Louis Children’s NICU, a major referral center with the highest attainable Level 4 designation. WashU Medicine physicians and St. Louis Children’s care team provide care for extremely premature infants and those with critical health issues in the state-of-the-art facility able to offer the most advanced treatments to ensure the best possible outcomes for these vulnerable babies.
Today’s careful use of antibiotics in preemies at St. Louis Children’s is grounded in a decadeslong, collaborative research effort among Warner and her WashU Medicine colleagues Phillip I. Tarr, MD, the Melvin E. Carnahan Professor of Pediatrics, and Gautam Dantas, PhD, the Conan Professor of Laboratory and Genomic Medicine. They pioneered the now widely accepted framework that harmful gut microbes are responsible for two major contributors to infant mortality: necrotizing enterocolitis (NEC), a devastating illness where bacteria invade the underdeveloped intestinal wall and cause tissue death, and bloodstream infections.
In a healthy full-term baby, billions of microbes move shortly after birth into the sterile gut and persist throughout life, influencing infection resistance, inflammation control and possibly long-term health. Birth, feeding and caregiver contact promote the healthy development of this microbial community.
But the WashU Medicine team has found that this process is disrupted in preemies who developed NEC. They reported, in the Lancet in 2016, that preemies with NEC housed unhealthy gut microbes before symptom onset, while healthy babies harbored more beneficial gut microbes. More recently, they reported that the harmful microbes built up over time in patients with NEC diagnosed after 40 days of life but not in healthy babies. The findings could inform development of a diagnostic tool to prevent or treat NEC sooner.
Preemies are vulnerable to unhealthy gut microbes, in part due to an immature immune system and a leaky gut barrier. Antibiotics can make them even more susceptible by creating space in the gut for unhealthy microbes to move in and persist for a long time, increasing the risk of bloodstream infection and NEC, the researchers found. Their research has added to the body of evidence that has heightened awareness of not only the benefits but also the risks of antibiotics.
“The results of our studies clearly demonstrated that the microbes that survived antibiotic treatment in preemies were more likely to be pathogenic,” said Tarr. “While there is no doubt that the widespread use of antibiotics has saved the lives of many infants, our research pointed out the need for safeguards to prevent their overuse, which can lead to significant harm, contributing to a more balanced approach in antibiotic administration.”
Bloodstream infections include early- and late-onset sepsis, with late-onset sepsis occurring after the first three days of life and affecting up to half of extremely preterm infants — those born before 27 weeks. Leading one of the first nationally funded studies of the early gut microbiome in preemies, Tarr, Warner, and a collaborator detected pathogenic bacteria in the guts of preemies with late-onset sepsis days before they appeared in the blood, suggesting that the harmful bacteria can leak into the bloodstream from the gut.
When Dantas joined WashU Medicine in 2009, he brought expertise in sequencing microorganisms and their antibiotic resistance genes to the new collaboration with Warner and Tarr. Aided by a series of highly talented graduate students and postdoctoral researchers, these investigators determined that preemies given antibiotics at three NICUs in the U.S. — St. Louis Children’s, Norton Children’s Hospital in Louisville and the Oklahoma Children’s Hospital — had more harmful gut bacteria compared with healthy full-term babies who didn’t receive antibiotics. What’s more, their guts still contained disease-causing bacteria carrying antibiotic-resistance genes a year and a half after they left the NICU. In a subsequent study, the team also found that 60% of the preemies who developed bloodstream infections in the NICU had nearly identical disease-causing strains in their guts immediately before the bloodstream infection.
“Antibiotics save lives,” said Warner. “But we have learned they are not risk-free. Although challenging, it is critical that we reduce antibiotic exposure in this vulnerable population of infants. We are committed to this challenge.”
Today, clinical guidelines for the responsible use of antibiotics in preemies, particularly the type and duration of antibiotics, are part of standard NICU protocols. Research from WashU Medicine and other institutions helped pave the way for these changes and also made a lasting impact by training numerous scientists who are now faculty elsewhere and continuing this work in their own labs.

The future: faster infection diagnosis, drug risk assessment
A challenge to reducing antibiotic overuse in preemies is that infections have overlapping symptoms with other problems; for instance, both infections and lung immaturity cause babies to breathe fast or even stop breathing for short periods. Being able to detect infections earlier would enable clinicians to use more targeted treatments and help preserve antibiotics’ effectiveness.
Drew Schwartz, MD, PhD, an assistant professor of pediatrics who completed his postdoctoral training in Dantas’ laboratory, is working to do just that. Schwartz’s lab uses advanced, rapid, bedside, long-read sequencing techniques to rapidly identify disease-causing microbes in preemies’ fecal samples, which could reduce diagnosis time to just one or two days, rather than two to five days. This is significant because infants with a suspected infection are typically started on broad-spectrum antibiotics, which are used to kill many types of bacteria, until they get a confirmed diagnosis. Faster identification of invading microbes can shift treatment from broad-spectrum antibiotics to more targeted ones sooner, reducing overall exposure.
“We once thought that generously giving antibiotics would protect preemies from infections,” said Schwartz, a specialist in infectious diseases who treats patients at St. Louis Children’s. “Now, we understand that it’s safer to limit antibiotics to cases where they are clearly needed. With WashU Medicine’s expertise in advanced sequencing techniques, we’re working to identify such cases faster and more accurately.”
Recently, researchers from the Dantas lab, including Schwartz, found in a study published in Cell Host & Microbe that medications commonly given to preemies to support their lungs, kidneys and heart — and that are not thought to directly target microbes — also drastically change the makeup of the microbiome, allowing harmful bugs to infiltrate the gut ecosystem.
The researchers are developing tools for physicians to help them assess a drug’s risk of altering a child’s microbiome and the likelihood of harmful bacteria growth before prescribing a drug.
“We are now thinking beyond antibiotics,” said Dantas. “As we work to come up with algorithms to help physicians understand a drug’s risk of impacting the microbiome, we now know to include most of the drugs given to preemies.”
Saving lives with judicious antibiotic use
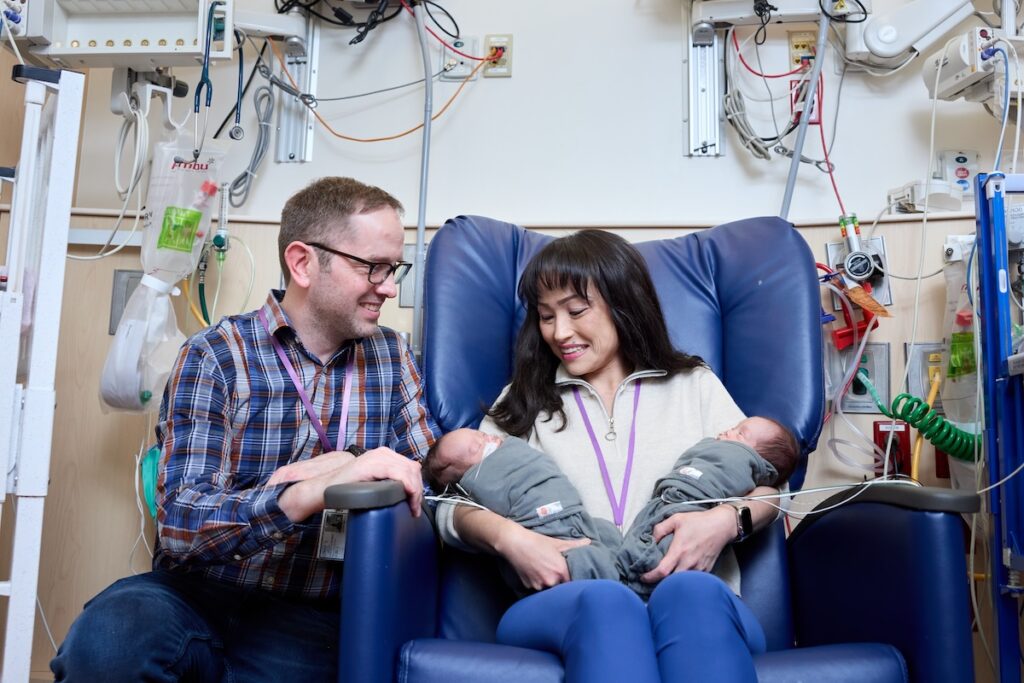
Linda Plummer knew she was experiencing a high-risk pregnancy. While expecting twins, she had been diagnosed by her obstetrician with an incarcerated uterus, a rare condition that traps the growing pear-shaped organ in the pelvis, threatening her pregnancy in the second trimester. During a visit with WashU Medicine’s maternal-fetal specialists, the doctors told Plummer and her husband Lucas Plummer to prepare for an imminent preterm birth.
Twins Livi and Logan were delivered via an emergency C-section after Livi’s heart rate became unstable. At less than 7 months’ gestation, Livi — 2 pounds, 8 ounces — and Logan — 3 pounds — were wheeled in incubators across a pedestrian bridge from Barnes-Jewish Hospital to the St. Louis Children’s NICU.
During their nearly 13- and 16.5-week stays, both babies, who were cared for by Warner, received antibiotics because their health conditions warranted the medications. Right away, Logan had trouble breathing, which improved after a course of antibiotics. While initially healthy, Livi’s health declined as her brother’s improved. At two weeks old, she developed NEC, which is fatal to roughly 30% of preemies who develop it.
The Plummers credit WashU Medicine doctors and St. Louis Children’s nurses and support staff for the prompt diagnosis and treatment that saved Livi’s life. A nurse spotted the faintest pink tinge of stool mucus in Livi’s diaper, and a subsequent abdominal X-ray caught the early stages of NEC. She was immediately treated with antibiotics and made a full recovery.
“At every juncture of our babies’ care, we had all the information to make a decision about giving them antibiotics,” said Linda Plummer. “Lucas and I said yes to antibiotics if the pros outweighed the cons, knowing that the information given to us by our clinical team is rooted in years of research performed right here.”
About WashU Medicine
WashU Medicine is a global leader in academic medicine, including biomedical research, patient care and educational programs with more than 3,000 faculty. Its National Institutes of Health (NIH) research funding portfolio is the second largest among U.S. medical schools and has grown 83% since 2016. Together with institutional investment, WashU Medicine commits well over $1 billion annually to basic and clinical research innovation and training. Its faculty practice is consistently among the top five in the country, with more than 2,000 faculty physicians practicing at 130 locations. WashU Medicine physicians exclusively staff Barnes-Jewish and St. Louis Children’s hospitals — the academic hospitals of BJC HealthCare — and Siteman Cancer Center, a partnership between BJC HealthCare and WashU Medicine and the only National Cancer Institute-designated comprehensive cancer center in Missouri. WashU Medicine physicians also treat patients at BJC’s community hospitals in our region. With a storied history in MD/PhD training, WashU Medicine recently dedicated $100 million to scholarships and curriculum renewal for its medical students, and is home to top-notch training programs in every medical subspecialty as well as physical therapy, occupational therapy, and audiology and communications sciences.
Originally published on the WashU Medicine website