
New research highlights how nerves – whether harmed by disease or traumatic injury – start to die, a discovery that unveils novel targets for developing drugs to slow or halt peripheral neuropathies and devastating neurodegenerative disorders such as Alzheimer’s disease, Parkinson’s disease and amyotrophic lateral sclerosis (ALS).
Peripheral neuropathy damages nerves in the body’s extremities and can cause unrelenting pain, stinging, burning, itching and sensitivity to touch. The condition is commonly associated with diabetes or develops as a side effect of chemotherapy.
The research, by scientists at Washington University School of Medicine in St. Louis, is reported online April 23 in the journal Science.
Nerve cells talk to each other by transmitting signals along communication cables called axons. Such signals underlie vital activities, such as thinking and memory, movement and language.
As part of the study, the researchers showed they could prevent axons from dying, a finding that suggests therapies could be developed to counteract the withering away of nerve axons.
“We have uncovered new details that let us piece together a major pathway involved in axon degeneration,” said senior author Jeffrey Milbrandt, MD, PhD, the James S. McDonnell Professor and head of the Department of Genetics. “This is an important step forward and helps to identify new therapeutic targets. That we were able to block axon degeneration in the lab also gives us hope that drugs could be developed to treat patients suffering from a variety of neurological conditions.”
A common thread among many neurological disorders and traumatic nerve injuries is the degeneration of axons, which interrupts nerve signaling and prevents nerves from communicating with one another. Axon degeneration is thought to be an initiating event in many of these disorders. In fact, an unhealthy axon is known to trigger its own death, and researchers are keenly interested in understanding how this happens.
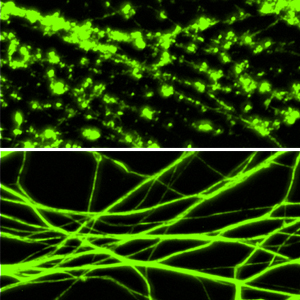
Working in cell cultures, fruit flies and mice, Milbrandt and co-author Aaron DiAntonio, MD, PhD, the Alan A. and Edith L. Wolff Professor of Developmental Biology, and their colleagues showed that a protein already known to be involved in axon degeneration, acts like a switch to trigger axon degeneration after an injury.
Moreover, they found that this protein, once unleashed, causes a rapid decline in the energy supply within axons. Within minutes after the protein – called SARM1 – is activated in neurons, a massive loss of nicotinamide adenine dinucleotide (NAD), a chemical central to a cell’s energy production, occurs within the axon.
“When a nerve is diseased or injured, SARM1 becomes more active, initiating a series of events that quickly causes an energetic catastrophe within the axon, and the axon undergoes self-destruction,” said first author Josiah Gerdts, an MD/PhD student in Milbrandt’s laboratory.
Working in neurons in which SARM1 was activated, the researchers showed they could completely block axon degeneration and neuron cell death by supplementing the cells with a precursor to NAD, a chemical called nicotinamide riboside. The neurons were able to use nicotinamide riboside to keep the axons energized and healthy.
Nicotinamide riboside has been linked in animal studies to good health and longevity, but its benefits have not been shown in people. The researchers said much more research is needed to know whether the chemical could slow or halt axon degeneration in the body.
“We are encouraged by the findings and think that identifying a class of drugs that block SARM1 activity has therapeutic potential in neurological disorders,” Milbrandt said. “The molecular details this pathway provides give us a number of therapeutic avenues to attack.”
The research is funded by the National Institutes of Health (NIH), grants RO1DA020812, RO1AG013730, RO1NS065053, RO1NS087632, RO1NS078007 and F31NS074517, and a grant from Vertex Pharmaceuticals.
Gerdts J, Brace EJ, Sasaki Y, DiAntonio A and Milbrandt J. SARM1 activation triggers axon degeneration locally via NAD+ destruction. Science. April 24, 2015.