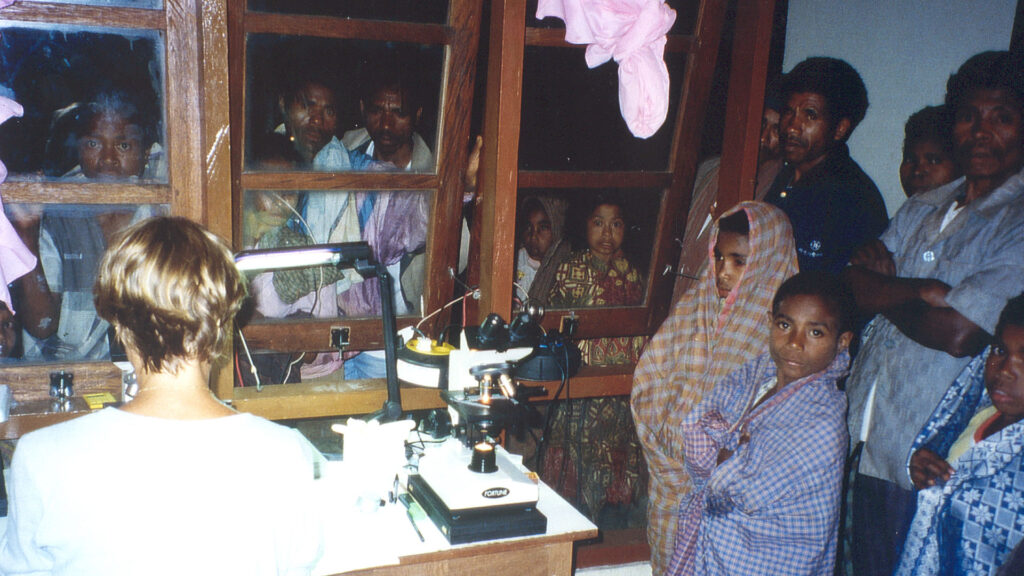
The parasites are active at night, so that’s when the researchers hunt them.
On the remote Indonesian island of Alor, Peter U. Fischer and his research collaborators use flashlights to set up makeshift labs.
On some parts of the island, they travel door to door in the hot, heavy night air — side-stepping aggressive dogs and fielding skeptical questions in the pursuit of human blood samples.
“It’s cumbersome and exhausting,” Fischer says. “But I’ve been doing it now for 30 years, so I’m used to it.”
WashU Medicine researchers work with local partners to reach residents in villages around the island.

On Alor Island in Indonesia, Peter Fischer, professor of medicine at WashU Medicine, collects blood samples. He and collaborators seek to determine whether one of three types of parasites that cause the disease lymphatic filariasis has been eliminated from the island.
Fischer, professor of medicine at WashU Medicine, heads the school’s Death to Onchocerciasis and Lymphatic Filariasis Project (DOLF). The program’s dramatic name feels appropriate given the audaciousness of its goal: the complete elimination of two devastating parasitic diseases. Lymphatic filariasis (LF, known as elephantiasis) and onchocerciasis (oncho, known as river blindness) cause illness and disability, affecting millions in tropical and subtropical regions of the world.
Thanks in part to DOLF-led research, progress has been tangible, though slower than initially hoped when the project launched in 2010 with funding from the Gates Foundation. A World Health Organization (WHO) target to eliminate LF by 2020 has come and gone.
“No new clinical cases have occurred in the last decade, but my impression doesn’t count for much. We need hard data to really show the absence of infection.”
Peter Fischer
Yet on Alor, Fischer and his collaborators see a welcome instance of hope and likely success. Following the mass distribution of medication in the mid-2000s, multiple research visits to the island point to the absence of Brugia timori, one of the three types of thread-like worms that cause LF.
“No new clinical cases have occurred in the last decade,” Fischer says, “but my impression doesn’t count for much. We need hard data to really show the absence of infection.”
In summer 2025, the researchers returned to the island with a single-minded question: Is B. timori gone for good?
The early picture
Fischer first made the multiday journey to Alor back in 2001, four years before joining the WashU faculty.
It was the first research trip that he and Kerstin Fischer, now a WashU Medicine senior scientist for DOLF, took as a married couple. (Last year, their grown son joined them.) Along with longtime collaborator Taniawati Supali and her team from the University of Indonesia, the scientists traveled to the island to assess the prevalence of LF. The presence of disease was devastatingly clear.


“I remember going to the market, and there were five women there,” Fischer says. “One of them had legs so swollen that she couldn’t walk.” They found a similar picture throughout the island. In addition to fever and headache, LF causes severe engorgement of the limbs. In men, it can also lead to swelling of the scrotum.
The researchers completed a study that confirmed the disease’s presence, as well as the culprits: the parasitic worms B. timori and Wuchereria bancrofti. Spread by the bites of infected mosquitos, the parasites had infected human hosts throughout the island. In the village of Mainang, 27% of nearly 600 tested individuals carried microfilariae, the first stage of parasite larvae.
Despite discovering such widespread disease, the researchers also saw a rare opportunity. Mainang and other inland, rice-farming villages on Alor were exclusively affected by B. timori. In comparison with other parasitic worms that cause LF, this one is relatively rare. It occurs only in East Nusa Tenggara (the Indonesian province that includes Alor) and Timor Leste, the island country to the south.
The process of field data collection has technologically become easier in the past two decades, Kerstin Fischer says. (Courtesy photo)
Given the isolated region and the smaller overall numbers of this parasite, the researchers believed in the possibility of complete eradication. While simultaneously pursuing numerous other DOLF projects around the globe, for years they’ve kept one eye on the tiny island.
To catch a parasite
To prevent the continued spread of LF, the WHO recommends preventive chemotherapy through mass drug administration (MDA).
This tactic provides donated medication once a year to entire communities where the disease is present. Since many of those who carry the parasites are asymptomatic, this broad approach offers the best chance of stopping further transmission.
These medications effectively kill microfilariae, the early-stage parasite larvae that circulate in an infected person’s bloodstream. But they don’t squash the adult worms that set up shop in their host’s lymphatic system. Once established, these worms wreak havoc, causing pain and stigma-inducing lymphedema. They also continue for years to produce more microfilariae by the thousands, which mosquitos can then transmit to more people.

To outlast the adult worms and their progeny, MDA programs typically require five years or more of annual treatment. More than 10 billion doses of MDA drugs targeting LF have been delivered since 2000, according to the WHO.
Though missing the original target to eliminate LF by 2020, these programs have alleviated tremendous suffering. By 2018, WHO’s Global Programme to Eliminate Lymphatic Filariasis had reduced the number of people living with the disease to 51 million — a massive number, to be sure, but a full 74% fewer cases than when the program launched in 2000.

(From left) Kerstin Fischer, Andreas Mating Ata, Tania Supali and Peter Fischer discuss Ata’s health and his experience with LF since he first met the researchers. The Fischers have collaborated for decades with Supali, and the team shares a longterm commitment to eliminating parasitic diseases.
Alor took part in one such effort, from 2002-06. The program used a then-novel two-drug treatment. Thanks to DOLF, an even more effective three-drug regimen, known by the acronym IDA, has since become WHO’s recommended standard in many parts of the world. Unlike earlier treatment combinations, IDA sterilizes or kills adult worms. This treatment has been tested for B. timori infection by Supali’s team on Indonesia’s Flores and Sumba islands.
Gary Weil, MD, professor emeritus of medicine, recently retired after founding DOLF and spending nearly 15 years at the project’s helm. In 2016, under Weil’s and Fischer’s watch, WashU Medicine led a randomized study on IDA, enrolling 26,836 participants across five LF-endemic countries: India, Papua New Guinea, Haiti, Indonesia and Fiji. The study proved that IDA was as safe as the two-drug cocktail.
One year later, the WHO formally reviewed these findings and endorsed the use of IDA for LF elimination. Since then, 300 million people in 22 countries have received the triple-drug treatment.
At a DOLF symposium in spring 2025 that brought international collaborators together, Weil recalled the early days of the project. “Progress was uneven in the LF elimination program, and the drugs were only partially effective,” he said.
“The Gates Foundation removed the excuse that many researchers have: ‘If only we had the money, we could do X, Y and Z.’ We were funded well. And to those whom much is given, much is expected.”
Gary Weil
The Gates Foundation wanted to change that, as did Weil and his collaborators on campus and around the world. Weil submitted a proposal for an extensive research project to improve treatments for LF and oncho, and the Foundation awarded funding in late 2009.
“The Gates Foundation removed the excuse that many researchers have: ‘If only we had the money, we could do X, Y and Z,’” Weil said at the symposium. “We were funded well. And to those whom much is given, much is expected.”
With concluded or ongoing study sites in 11 countries, the project continues to fulfill those expectations. DOLF efforts involve every stage of the MDA process — diagnostics, new treatment testing, implementation research and follow-up surveillance. The public health implementation component is extremely important, Fischer says, because the medications do no good if people won’t swallow the pills.
Much of DOLF’s work pursues site-specific findings aimed at refining MDA strategies — because when combating global diseases, the best treatment for one area may prove ineffective, or even damaging, in another.
For example, IDA treatment cannot be used in many parts of Africa. In areas where LF exists alongside diseases like oncho, which affect the eyes, the three-drug combination could cause vision loss or other serious health problems. Meanwhile, in other parts of the world, the two-drug option has proved sufficient. The DOLF researchers had hopes that this was true for Alor.
Alor today
When Tania Supali, Peter Fischer and Kerstin Fischer arrived on the island last summer, visual signs of LF were practically nonexistent.
A few older residents bore signs of past infection, with swelling that never fully resolved after years of earlier assault to their lymphatic systems. But many living on the island, especially younger people, had never even heard of LF.
The absence of suffering was gratifying to the researchers, who have worked for so long to bring about similar results around the globe. However, for their current aim — data collection to determine the presence or absence of B. timori — the lack of widespread symptoms led to a challenge.

Lymphatic filariasis can cause fluid buildup in the limbs. In some patients, like Martha Mating Ata (left), the swelling persists for years.
“It’s good for our purpose, but it makes it scientifically difficult to get enough samples,” Peter Fischer says. “Sometimes many people wanted to give a blood sample. But in other cases, we had difficulties.”
Hence the door-to-door visits after dark. In places with plenty of daytime volunteers, the researchers ran quick antibody tests on participants, with positive results indicating either a previous or current infection. Those with positive results were asked to return at night for a follow-up test to see whether live B. timori still resided in their blood. In areas with few daytime participants, however, the researchers had to venture out into neighborhoods at night to seek out samples.
Why the cover of darkness? As with other filarial parasites, B. timori matches the activity patterns of the mosquito vectors that help it spread. While human hosts sleep, microfilariae wriggle in peripheral blood, waiting to be picked up by a passing hungry mosquito. Inside the mosquito, the microfilariae then develop into mature, infective larvae that are ready to be deposited into another host.
The nighttime activity of filarial worms encourages transmission. It also allows for the microfilariae to be detected in a finger-prick blood smear with a microscope. The scientists just need to be in the right place, and at the right hour.


In some ways this process of nighttime collection and analysis has become easier over the years, Kerstin Fischer says. Barcodes have replaced hand-written labels to keep track of samples. Scientific equipment can be obtained in Jakarta, so the WashU Medicine team no longer must pack every item from home laboratories. Fischer used to agonize about how many pipette tips and other supplies to bring on trips like these, and that is less of an issue now. But challenges remain in field data collection.
“When you are working in these environments, there’s a lot of humidity,” she says. “If you’re doing blood filtration, for example, you’re supposed to wear goggles and your lab coat and gloves, but the gloves are wet inside after five minutes. Sometimes even your goggles fog up, and you can’t really work with foggy goggles.”
Fischer has conducted similar work in Liberia, Ghana and Uganda. Even though the conditions on Alor made the science challenging, it was a reminder of some of her favorite parts of her career.

WashU Medicine researchers visit a market on Alor Island. When they first arrived more than 20 years ago, signs of LF were prevalent throughout the island. In 2025, very few people bore signs of the disease.
“The people who I met were so friendly,” Fischer says. “Sometimes you forget when you go into these countries, into these villages, that there’s so much laughter. There is the everyday work, which is so hard, but there is also community.
“This is why I chose to be in the field,” she says. “I love field work.”
After a week of work, the group had screened around 1,200 individuals, Peter Fischer says. In Mainang, 328 people took the antibody screening test; only 14 tested positive. A greater percentage of tests in the village of Alemba yielded positive results. There, 35 out of 294 tested adults carried the B. timori antibodies.
Because antibodies can linger long after the death of disease-causing parasites, the team still held hope that the worms were gone.
At night, the antibody-positive individuals came back for follow-up tests. One by one, their results came back — negative after negative. The researchers found zero cases of B. timori microfilariae on Alor.
“We haven’t seen any filaria larvae in any individuals who were screened,” Fischer says. “We didn’t see any evidence that there is still transmission going on and that there are still parasites around.

Alor resident Martha Mating Ata is one of many in the region who has suffered from LF.

“These are very promising results,” he continues. “It’s the first step toward documenting that this filarial species, B. timori, may be eliminated. In addition, we did not find any W. bancrofti microfilariae in 602 adults from two coastal villages.”
A meticulous scientist, Fischer won’t claim victory yet. The WHO has strict criteria that must be met to certify that a country is free of LF, but no such formal guidance exists for confirming the elimination of a pathogen species that causes it. His team is seeking direction on what evidence the WHO would need to make such a declaration. They hope that the encouraging results from Alor will garner support to test additional districts in Indonesia that were formally endemic for this worm.
“You can’t put a number on the elimination of a pathogen. The elimination is priceless. All people, all children who are born now, are protected.”
Peter Fischer
Fischer acknowledges that the elimination of B. timori is a small part of the overall fight against LF, oncho and other filarial diseases. But whatever is needed to confirm eradication of one disease-causing pathogen, the team plans to do.
“People sometimes ask: ‘Why is this important? What is the real value?’” Fischer says. “And the answer is that you can’t put a number on the elimination of a pathogen. The elimination is priceless.
“All people, all children who are born now, are protected. They won’t get sick again from B. timori.”
The journey continues
This moment of good news sustains DOLF researchers as they pursue the full elimination of LF on other fronts.
Progress is rarely straightforward. The Gates Foundation continues to fund DOLF efforts in another part of Indonesia, for example, where recent findings are less celebratory. While B. timori is on the run, or perhaps gone for good, a more common LF-causing parasite, B. malayi, has been hiding out — in monkeys.
Thousands of miles west of Alor, Indonesia’s Belitung District also carried out five rounds of MDA, completed in 2010. Some WHO-recommended follow-up surveys suggested success. But in 2021, additional surveys proved that LF had snuck back into the region. Around 2% of those tested across five villages carried microfilariae.
In collaboration with Tania Supali and her team, Irina Diekmann, a veterinarian and DOLF postdoctoral researcher, visited Belitung in 2023 to find out if B. malayi was present in local animals, as well as in humans. The group tested blood from 291 cats, 41 dogs and 163 crab-eating macaques (a type of monkey). The science was successful but the results discouraging: Among the monkeys, they found B. malayi DNA in blood from 13% of those tested.
The existence of an animal reservoir greatly complicates future treatment plans. After all, monkeys do not readily participate in MDA programs. But Fischer and his collaborators emphasize that even when tactics must change, the work continues. On Belitung, future efforts must now also target the specific types of mosquitos that carry the pathogen from monkeys to humans and back again.
Elsewhere in the world, DOLF researchers continue to pursue more effective diagnostics, medications and methods.
The director of clinical research for DOLF, Philip Budge, MD, PhD, authored a study last year showing that the medication moxidectin is more effective and longer lasting for treating LF than ivermectin, one of the three essential drugs in IDA. Moxidectin is already approved to treat oncho.
“Moxidectin really works much better than the drugs that we’re currently using against lymphatic filariasis,” says Budge, associate professor of medicine at WashU Medicine. “If you treat someone with moxidectin, they are more likely to clear their parasites for longer. So, maybe the right place for moxidectin in the global elimination program is in people who are hard to reach repeatedly.”
Enacting this vision for moxidectin would require the largesse of pharmaceutical companies. In 1987, P. Roy Vagelos, MD, a former WashU Medicine faculty member and renowned physician-scientist who was then CEO and chairman of Merck & Co., spearheaded the long-term donation of ivermectin for MDA efforts. The pharmaceutical company GSK donates albendazole, another essential LF drug.
Moxidectin, however, has no such large-scale donor, Fischer says. Though the drug is synthesized in large quantities for the veterinary market, for the treatment of heartworms, human pharmaceutical standards are much different.
In addition to natural setbacks, like the existence of an animal reservoir in Indonesia, human-caused setbacks, like massive cuts to funding from the U.S. Agency for International Development (USAID), also strain the research community’s dogged efforts to rid the world of LF and similar diseases. The WHO estimates that 47 MDA programs for 150 million people were halted by the cuts.
At the DOLF symposium last year, a panel discussed the funding issues. Though the interruptions will push back the timeline of LF elimination in many areas, the experts aim to help countries absorb the responsibilities of MDA administration into their own public health systems.
International collaboration will be key. Among DOLF researchers, it’s already the standard. The symposium alone welcomed 80 participants from 15 countries, all of whom are committed to the elimination of LF, oncho and related diseases.
“It takes time, and it takes people who are devoted to the project as you are,” Fischer says. For the work in Alor and across Indonesia, Fischer is referring to Supali and her team, who have been on the ground for decades.
“Tania and her team, they are as devoted to elimination as I am,” Fischer says. “To be able to do this over such a long period of time is only possible if you have support.”
Support from international and campus partners. Support from the Gates Foundation. Support from pharmaceutical companies, the Barnes Jewish Hospital Foundation and other organizations. And together, a joint determination to eliminate pathogens and improve lives.
“For many diseases, we are not quite there yet to eliminate a disease or eliminate pathogens,” Fischer says. “But for lymphatic filariasis, we have the tools.”