In some ways a cell in your body or an organelle in that cell is like an ancient walled town. Life inside either depends critically on the intelligence of the gatekeepers.
If too many barbarians sneak into town, the town may be put to the torch. And if the cellular gatekeepers can’t control the flow of ions and molecules into and out of the cell, the cell may die.
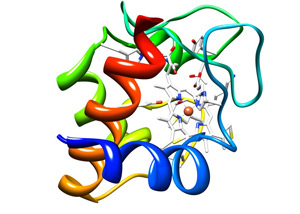
Resource for Biocomputing, Visualization, and Informatics at the University of California, San Francisco
The cytochrome c protein (colored ribbons), holds in its embrace a heme group (white honeycomb) that in turn clasps an iron atom (orange ball). This molecule is essential to life and any chemical that interferes with its activity is lethal.
Because of their importance, cellular gates, channels and transporters, are the targets of intense scientific interest.
One substance that has to cross the cell membrane is a molecule called heme, which plays a crucial role in supplying the cell with the energy needed to carry out the chemical reactions that sustain life.
Although heme is made inside a bacterium or cell organelle, it must be moved outside and plugged into a protein before it becomes functional. What’s more, because it is chemically vulnerable, it must be protected as it makes this mini-journey.
Earlier this year scientists at Washington University in St. Louis discovered a channel present in plants and many bacteria that both transfers and protects the heme. The work, done by graduate student Elaine Frawley and biology professor Robert Kranz, was recently published in the Proceedings of the National Academy of Sciences
What’s a heme when it’s at home?
Heme is a circlet of atoms that hold an iron atom much as a setting holds the diamond in a ring.
The body needs iron because it easily accepts and donates electrons, which makes it useful for all sorts of cellular chemistry. But, by the same token, free iron would participate in destructive chemical reactions whose byproducts might kill cells. This is one reason iron is usually bound in heme or in other proteins.
Heme is the essential component of two types of molecules. One is the hemoglobin molecules that bind oxygen in the lungs and ferry it around the body.
More than half the heme in the body is in hemoglobin, but some of the rest is in the hemeproteins called cytochromes. These are part of a chain of molecules embedded in the membranes of cell organelles that wring energy out of food.
The molecules in the chain extract electrons from sugar molecules, siphoning off a little of their energy, and passing them on to the next molecule, which also siphons off a little energy, and so on.
Kranz has devoted much of his career to understanding the molecular machinery that makes cytochrome c, one of the most important of these proteins. There are several different versions of this machinery, but the heme group is always assembled in the protected environment inside a cell organelle or bacterium, and then moved outside, where it is locked into a cytochrome c protein sitting on the outside of the cell membrane.
Trapping the heme in the channel protein
To figure out how heme gets across the membrane, Frawley used a benign strain of common gut bacterium Escherichia coli. But this was a special E. coli whose own cytochrome-c making machinery had been removed and replaced with machinery taken from Helicobacter hepaticus, a recently discovered bacterium that can cause hepatitis.
The Helicobacter system for making cytochrome features a humongous protein called CcsBA that threads repeatedly through the cell membrane, leaving messy loops on either side like the stitches made by a sewing machine whose tension is out of adjustment. The parts of the molecule that pass through membrane are called transmembrane domains.
By trapping the heme inside the isolated channel protein, Frawley showed that two transmembrane domains come together to form a channel for the heme.
Proteins like CcsBA that are embedded in the cell membrane are difficult to work with, Kranz explains. The membrane is a lipid, or fat, so trying to extract a membrane protein is like trying to wash greasy dishes in cold water. Frawley had to use detergents to separate the protein, he says, and even then it took her a year or two to purify enough protein to work with.
But then there was a eureka moment. “When I got enough pure protein and concentrated it in the test tube, I could see it was tinted red, the color of heme,” Frawley says. That told me I had trapped the heme in the channel protein. I was ecstatic!”
Just to make sure her eyes weren’t fooling her, she analyzed the sample spectroscopically and confirmed that it was indeed absorbing light at the wavelengths characteristic of heme.
Heme guards
But that isn’t the end of the story. The heme is just a building block, a part rather than a whole. Like the parts of a lamp or table that you buy in a cardboard box and have to assemble yourself, it has “projections” that need to be protected until you can stick them into the corresponding “holes” in another part.
The “projections” are available electrons, and the problem with heme is to prevent it from oxidizing, or giving up some of its spare electrons, before it can be plugged into the waiting cytochrome. Only the non-oxidized form can form the correct bonds with the cytochrome precursor on the outside of the cell. But while it is traversing the channel and before it is bound to the cytochrome, the heme is exposed to a hostile, oxidizing environment.
How is it protected in transit? Frawley and Kranz knew that some parts of the membrane protein CcsBA are highly conserved, meaning that no matter how evolution alters the rest of the protein, it leaves these sections alone. They suspected the conserved bits, four copies of the amino acid histidine, are guard molecules that protect the heme from oxidizing.
When Frawley mutated the histidines on the inside of the membrane, the protein’s absorption spectrum told her it had stopped functioning. The heme channel protein couldn’t bind the heme and shuttle it through to the outside anymore.
“That’s the key point of the paper,” says Kranz. “There’s a heme channel and the histidines have to be there to both bind heme and protect the heme from the environment.”
To prove the mutated histidines were the problem, she added imidazole, a small compound that is chemically the same as part of the histidine, to her E. coli cultures. The imidazole fixed the broken channel and the E. coli started making cytochrome again.
The imidazole trick, Kranz says, “is probably the coolest result I’ve had in 23 years in my lab.”
For more information about Robert Kranz’s research, click here.